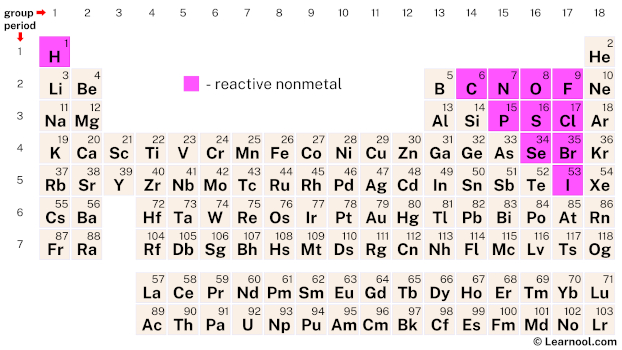
This study may also determine all those precious metals which are not functionally reused, and will collect information on finish-of-lifestyle failures, exploration companionality, and transfer dependency. Inside the perspective of the recycling of materials as well as their components, a lot of intricate metallic alloys make up five or higher elemental ingredients. Metals are grouped according to their positions in the periodic table, because of their properties. Moreover, these factors are susceptible to drop electrons, and will kind ions having a good demand or no demand by any means. /complete-periodic-table-of-elements-royalty-free-vector-166052665-5a565f0e47c2660037ab8aca.jpg)
Materials display different qualities, which include electric powered conductivity, lustre, and malleability. This group of people consists of each materials and nonmetals. The primary number of the periodic kitchen table includes the elements with s and p electron designs. The properties of move precious metals are essential for his or her utilization in environmental surroundings, in addition to their unique properties also assist to clarify their reputation amongst researchers. Their most affordable boiling and melting things are connected with no-alloys. The greatest boiling and melting points are related to transition metals. These reactive elements typically develop ores with some other metals and no-alloys. In the Periodic Desk, move materials are in a natural way plentiful aspects within the earth's crust. In the Periodic Table, alkali metals are classified as the s-prevent factors. They also become more reactive in water, as their reactivity increases. They can be typically present in salts, and also have a physique-focused cubic structure. These metals are most reactive when in contact withwater and air. The label alkali metals originates from the Arabic phrase al-qali, significance ashes. Most Reactive Metal In Group 2 Of The Periodic Table. These alloys are normally used for making powerful resources, for example stainlesss steel and metal, and they are generally less reactive than other factors. You may also learn about changeover precious metals, a team of metals that drop between your major groups. Moreover, we'll include several of the more complex metallic alloys, which include gold and silver. They conduct electricity in molten state.Most Reactive Metal In Group 2 Of The Periodic Table - There are numerous sets of alloys in the Occasional Dinner table, and this post will investigate the primary categories of these components. They have high melting and boiling points.Ĥ. The resulting compound is called an ionic compound.Ģ. Therefore, when a metal reacts with a non-metal, a transfer of electrons takes place, resulting in the formation of an ionic bond. Metals are electropositive and have a tendency to lose electrons while non-metals are electronegative and have a tendency to accept electrons. Halogens are the non-metal elements which belong to group 17 of the periodic table. The following drawing clearly explains the reaction of Sodium and Fluorine: When Sodium and Fluorine react, a transfer of electrons takes place from Sodium to Fluorine and an ionic compound is formed. 
Sodium has 1 electron in its outermost shell whereas Fluorine has 7 electrons. Let us see the formation of an ionic compound, namely Sodium Fluoride (NaF), when Sodium (Group 1 element) reacts with Fluorine ( Group 17 element). Sodium (Na) and Potassium (K) are the two very reactive metals belonging to group 1 of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
